|
Intracellular pathogens present a
significant challenge for vaccine development as these agents invade
host cells and are able to avoid initial recognition by the host immune
system. Intracellular pathogens often multiply undetected until
cytotoxic T-lymphocytes (CTL) recognize foreign antigenic peptides
displayed in association with surface MHC class I molecules and are
activated to kill the infected host cell.
Listeriolysin O (LLO) is a secreted
pore-forming cytolysin that mediates vacuole escape by L. monocytogenes. I have been
working with a modified
version of LLO that retains full cytolytic activity
but is not
secreted. L. monocytogenes expressing this cytoplasmic LLO
(cytoLLO) are unable to escape the vacuole and do not replicate within
host
cells. However, when these bacteria are eventually degraded within
phagosomes, active cytoLLO is released and perforates the
phagosomal membrane. Antigens from the degraded bacteria can then enter
the cytosol and be processed for presentation on host cell MHC class I
molecules. CTL can then recognize displayed antigenic peptides and
generate an immune response.
I am further investigating the use
of cytoLLO-expressing bacteria as a safe and
effective vaccine strategy against intracellular pathogens.
View Larger Image
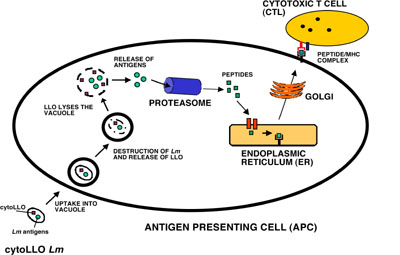
Figure.
CytoLLO Listeria monocytogenes
(Lm) is taken up by an antigen
presenting cell (APC) and degraded in the vacuole. Released cytoLLO
lyses the vacuole and allows access of Lm antigens to the cytosol of the
APC. The released antigens are processed by the proteasome into
peptides that are transported to the endoplasmic reticulum (ER). In the
ER, peptides are assembled onto MHC Class I molecules. The peptide/MHC
complex is trafficked via the Golgi to the surface of the APC where it
can be engaged by a cytotoxic T cell (CTL).
|
